Technology
COLLPLANT BIOTECHNOLOGIES REPORTS 2024 THIRD QUARTER FINANCIAL RESULTS AND PROVIDES CORPORATE UPDATE
Published
1 year agoon
By
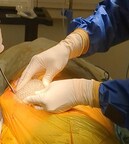
-Promising results obtained from the pre-clinical study with CollPlant’s commercial sized rhCollagen-based regenerative breast implants, demonstrating significant implant vascularization and rapid ingrowth of native tissue
-Innovative breast implant technology designed to address a $3.0 billion market opportunity
-Cash and cash equivalents balance as of September 30, 2024 was $15.4 million
-Conference call to be held today at 10:00 a.m. U.S. EDT –
REHOVOT, Israel, Nov. 27, 2024 /PRNewswire/ — CollPlant Biotechnologies (Nasdaq: CLGN), a regenerative and aesthetics medicine company developing innovative technologies and products based on its non-animal-derived collagen for tissue regeneration and medical aesthetics, today announced financial results for the third quarter ending September 30, 2024, and provided a corporate update.
“We are now one step closer to advancing our breast implant program into human studies, after developing a biocompatible commercial-sized, 3D-printed implant with a natural feel that has shown promising results in preclinical trials,” commented Yehiel Tal, Chief Executive Officer of CollPlant Biotechnologies. “So far, the study is showing encouraging outcomes three months post implantation, with evidence of significant implant vascularization and rapid ingrowth of native tissue, both of which are critical factors in enabling effective integration of the implant with the physiological system and supporting long-lasting regenerative processes. We are looking forward to reporting additional results from this program in the first quarter of 2025. “
Mr. Tal, continued, “We have recently adjusted our development and operation plans so that the Company is capitalized for a period of at least one year from today. In addition, we are prioritizing raising non-dilutive cash through the creation of additional collaborations and to that end we are engaging in dialog with companies in the medical and aesthetics fields that have interest in our rhCollagen technology. “
Dr. Sachin M. Shridharani, board-certified Plastic Surgeon, founder of LUXURGERY© New York and Associate Clinical Professor of Plastic Surgery at Washington University School of Medicine added, “CollPlant’s novel, 3D-printed, biocompatible and regenerative breast implant technology is an incredibly exciting development with multiple applications. There is an unmet need in aesthetic and reconstructive plastic surgery for highly biocompatible materials to be employed for breast augmentation and reconstruction. Eliminating immune responses to materials implanted in the breast or any portion of the body serves as a holy grail in plastic surgery.”
Q3 and Recent Program Highlights
In August, CollPlant announced the launch of a pre-clinical study with 200cc commercial-sized breast implants printed using CollPlant’s bioinks and Stratasys’ (Nasdaq: SSYS) Origin 3D printer. The collaboration between CollPlant and Stratasys has been focused on the development of a bioprinting solution for CollPlant’s regenerative breast implants. In addition, the companies aim to develop solutions for scaling-up the implant bioprinting process. If successfully developed, the novel implants could provide a revolutionary alternative to the implants that are currently on the market.
To date, the study has shown promising results three months post implantation, demonstrating significant implant vascularization and rapid ingrowth of native tissue, both of which are critical factors in enabling effective integration of the implant with the physiological system and supporting long-lasting regenerative processes.
Collaboration Updates
The collaboration with AbbVie continues to develop a dermal and soft tissue filler product for the medical aesthetics market. According to the agreement between the parties, CollPlant granted AbbVie worldwide exclusive rights to use its rhCollagen for the production and commercialization of a final dermal filler product. In return, CollPlant is entitled to receive, among other terms, up to $50 million and a fixed-fee royalty payment for each product commercialized. As of today, $24 million has been paid to CollPlant. AbbVie is responsible for all the costs of the dermal and soft tissue filler product candidate development, including the costs of clinical trials.On November 11, 2024, CollPlant presented at the ISBF (International Society for Biofabrication) 2024 conference. ISBF is a professional society promoting biofabrication research and development for medical applications. During the conference, CollPlant demonstrated its technology platform for mass production of human collagen and biofabrication of its innovative regenerative breast implants.
On July 29, 2024, CollPlant announced the release of its inaugural Environmental, Social and Corporate Governance (ESG) and Sustainability Report covering the fiscal year 2023. The report reflects CollPlant’s wide commitment to fostering environmental sustainability and enhancing human health, as well as advancing social and corporate governance objectives that contribute to the Company’s impact.
Three- and Nine-Month-Period Ended September 30, 2024 Financial Results
GAAP revenues for the third quarter ended September 30, 2024, were $4,000 compared to $43,000 for the third quarter ended September 30, 2023. The decrease in revenues is mainly related to the decrease in sales of rhCollagen to the Company’s largest customer. The deliveries to this customer are in accordance with the development plan of the customer, and in the third quarter no deliveries were planned or made. In accordance with the plan, CollPlant is prepared to supply rhCollagen to this customer this quarter, during December 2024.
GAAP revenues for the nine months ended September 30, 2024, were $351,000 compared to $10.7 million for the nine months ended September 30, 2023. The decrease of approximately $10.3 million is related to the achievement of a milestone in 2023, which triggered a $10 million payment under the AbbVie Agreement, as well as an approximate $300,000 decrease in sales of rhCollagen and VergenixFG.
GAAP cost of revenues for the third quarter ended September 30, 2024, was $272,000, compared to $278,000 for the third quarter ended September 30, 2023.
GAAP cost of revenues for the nine months ended September 30, 2024, was $1.4 million, compared to $1.2 million for the nine months ended September 30, 2023. The increase in cost of revenues in the amount of approximately $200,000 mainly comprised of (i) a $452,000 increase related to inventory impairment, offset by (ii) a decrease of $308,000 in royalty expenses to the IIA, mainly related to the milestone payment received from AbbVie in 2023.
GAAP gross loss for the third quarter ended September 30, 2024, was $268,000, compared to $235,000 in the third quarter ended September 30, 2023.
GAAP gross loss for the nine months ended September 30, 2024, was $1.0 million, compared to gross profit of $9.4 million for the nine months ended September 30, 2023.
GAAP operating expenses for the third quarter ended September 30, 2024, were $4.3 million, compared to $4.4 million in the third quarter ended September 30, 2023. The decrease of approximately $100,000 is mainly related to (i) a decrease of $213,000 in employees’ salaries expense and related share-based compensation expenses, offset by (ii) an increase of $358,000 in research and development activities mainly related to the breast implants program. On a non-GAAP basis, operating expenses for the third quarter ended September 30, 2024 were $3.8 million, compared to $3.9 million for the third quarter ended September 30, 2023. Non-GAAP measures exclude certain non-cash expenses.
GAAP operating expenses for the nine months ended September 30, 2024, were $12.3 million, compared to $11.9 million for the nine months ended September 30, 2023. The increase of approximately $400,000 is mainly related to an increase in research and development activities mainly related to the breast implants program. On a non-GAAP basis, operating expenses for the nine months ended September 30, 2024, were $11.0 million, compared to $10.6 million for the nine months ended September 30, 2023.
GAAP financial income, net, for the third quarter ended September 30, 2024, totaled $216,000, compared to $225,000 in the third quarter ended September 30, 2023.
GAAP financial income, net, for the nine months ended September 30, 2024, totaled $546,000, compared to $114,000 for the nine months ended September 30, 2023. The increase in financial income is due to interest received from the Company’s short-term cash deposits and exchange rate differences.
GAAP net loss for the third quarter ended September 30, 2024, was $4.3 million, or $0.38 basic loss per share, compared to a net loss of $4.4 million, or $0.38 basic loss per share, for the third quarter ended September 30, 2023. Non-GAAP net loss for the third quarter ended September 30, 2024, was $3.8 million, or $0.33 loss per share, compared to a net loss of $4.0 million, or $0.35 basic loss per share, for the third quarter ended September 30, 2023.
GAAP net loss for the nine months ended September 30, 2024, was $12.7 million, or $1.11 basic loss per share, compared to a net loss of $2.3 million, or $0.2 basic loss per share, for the nine months ended September 30, 2023. Non-GAAP net loss for the nine months ended September 30, 2024, was $11.5 million, or $1.0 loss per share, compared to a net loss of $1.2 million, or $0.11 basic loss per share, for the nine months ended September 30, 2023.
Balance Sheet and Cash Flow
Cash and cash equivalents as of September 30, 2024 were $15.4 million. The cash balance represents a company cash runway that will satisfy the Company’s operations requirements at least until the end of 2025, based on currently contemplated operations and plans. If the Company does not obtain additional funding sources when necessary to support its cost structure, it will implement cost reduction measures. These plans may include organizational adjustments and additional cost reductions if needed.
Cash used in operating activities during the nine months ended September 30, 2024, was $10.6 million compared to $418,000 during the nine months ended September 30, 2023.
Cash used in investing activities during the nine months ended September 30, 2024, was $481,000 compared to $784,000 during the nine months ended September 30, 2023 and related primarily to the purchases of property and equipment.
Cash provided by financing activities during the nine months ended September 30, 2024 was $9,000 compared to $1.1 million during the nine months ended September 30, 2023.
Conference call information
To participate in the conference call, please use the dial-in information below:
U.S. investors: 1-877-407-9716
Investors outside of the U.S.: 1-201-493-6779
Israel investors: 1-809-406-247
Conference ID: 13749096
Note, you can avoid long wait times for the operator by using the Call me™ feature and clicking the link below 15 minutes prior to the scheduled call start time:
https://callme.viavid.com/viavid/?callme=true&passcode=13728588&h=true&info=company-email&r=true&B=6
Webcast information
A live webcast will also be available in listen-only mode and can be accessed here or via the link to be posted on the News & Events section of the CollPlant Investor relations website. A replay of the webcast will be available following the conclusion of the live broadcast and will be accessible on the Company’s website for a limited time.
Submit questions to management in advance of the call
To ask management a question ahead of the call, please email Dan Ferry at LifeSci Advisors LLC up until 24 hours before the event at daniel@lifesciadvisors.com.
COLLPLANT BIOTECHNOLOGIES LTD.
CONDENSED CONSOLIDATED BALANCE SHEETS
(U.S. dollars in thousands)
September 30,
December 31,
2024
2023
(Unaudited)
Assets
Current assets:
Cash and cash equivalents
$
15,371
$
26,674
Restricted deposit
241
241
Trade receivables, net
5
–
Inventories
454
714
Other accounts receivable and prepaid expenses
426
393
Total current assets
16,497
28,022
Non-current assets:
Restricted deposit
115
57
Operating lease right-of-use assets
3,225
3,070
Property and equipment, net
2,460
2,789
Intangible assets, net
145
188
Total non-current assets
5,945
6,104
Total assets
$
22,442
$
34,126
COLLPLANT BIOTECHNOLOGIES LTD.
CONDENSED CONSOLIDATED BALANCE SHEETS
(U.S. dollars in thousands, except share data)
September 30,
December 31,
2024
2023
(Unaudited)
Liabilities and shareholders’ equity
Current liabilities:
Trade payables
$
1,009
$
980
Operating lease liabilities
798
624
Accrued liabilities and other
1,262
1,647
Total current liabilities
3,069
3,251
Non-current liabilities:
Operating lease liabilities
2,455
2,535
Total non-current liabilities
2,455
2,535
Total liabilities
5,524
5,786
Commitments and contingencies
Shareholders’ Equity:
Ordinary shares, NIS 1.5 par value – authorized: 30,000,000 ordinary shares as of
September 30, 2024 (unaudited) and December 31, 2023; issued and outstanding:
11,454,512 and 11,452,672 ordinary shares as of September 30, 2024 (unaudited)
and December 31, 2023, respectively
4,983
4,982
Additional paid in capital
122,376
121,068
Accumulated other comprehensive loss
(969)
(969)
Accumulated deficit
(109,472)
(96,741)
Total shareholders’ equity
16,918
28,340
Total liabilities and shareholders’ equity
$
22,442
$
34,126
COLLPLANT BIOTECHNOLOGIES LTD.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(U.S. dollars in thousands, except share and per share data)
(Unaudited)
Nine months ended
September 30
Three months ended
September 30
2024
2023
2024
2023
Revenues
$
351
$
10,660
$
4
$
43
Cost of revenues
1,353
1,218
272
278
Gross profit (loss)
(1,002)
9,442
(268)
(235)
Operating expenses:
Research and development
7,972
7,371
2,869
2,695
General, administrative and marketing
4,303
4,515
1,405
1,672
Total operating loss
(13,277)
(2,444)
(4,542)
(4,602)
Financial income, net
546
114
216
225
Net loss for the period
$
(12,731)
$
(2,330)
$
(4,326)
$
(4,377)
Basic and diluted net loss per ordinary share
$
(1.11)
$
(0.20)
$
(0.38)
$
(0.38)
Weighted average ordinary shares
outstanding used in computation of
basic and diluted net loss per share
11,454,069
11,367,767
11,454,512
11,443,023
COLLPLANT BIOTECHNOLOGIES LTD.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(U.S. dollars in thousands)
(Unaudited)
Nine months ended
September 30
2024
2023
Cash flows from operating activities:
Net loss
$
(12,731)
$
(2,330)
Adjustments to reconcile net income (loss) to net cash used in operating activities:
Depreciation and amortization
796
818
Share-based compensation to employees and consultants
1,295
1,373
Net loss from financing expenses
201
568
Changes in operating asset and liability items:
Decrease (increase) in trade receivables
(5)
5
Decrease (increase) in inventories
265
(107)
Decrease (increase) in other accounts receivable and prepaid expenses
(33)
50
Decrease in operating right of use assets
468
387
Increase (decrease) in trade payables
29
(389)
Decrease in lease liabilities
(529)
(669)
Decrease in accrued liabilities and other payables
(385)
(124)
Net cash used in operating activities
(10,629)
(418)
Cash flows from investing activities:
Purchase of property and equipment
(424)
(725)
Investment in restricted deposits
(57)
(59)
Net cash used in investing activities
(481)
(784)
Cash flows from financing activities:
Exercise of options and warrants into shares
9
1,108
Net cash provided by financing activities
9
1,108
Exchange differences on cash and cash equivalents and restricted cash
(202)
(578)
Net decrease in cash and cash equivalents and restricted cash
(11,303)
(672)
Cash and cash equivalents and restricted cash and at the beginning of the period
26,674
29,653
Cash and cash equivalents and restricted cash at the end of the period
$
15,371
$
28,981
COLLPLANT BIOTECHNOLOGIES LTD.
APPENDICES TO CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(U.S. dollars in thousands)
(Unaudited)
Nine months ended
September 30
2024
2023
Appendix to the statement of cash flows
A. Supplementary information on investing and financing activities not involving
cash flows:
Right of use assets recognized with corresponding lease liabilities
$
623
$
855
Capitalization of Share-based compensation to inventory
$
5
$
34
B. Reconciliation of Cash and cash equivalents at the end of the period
Cash and cash equivalents
$
15,371
$
28,981
Total cash and cash equivalents
$
15,371
$
28,981
COLLPLANT BIOTECHNOLOGIES LTD.
RECONCILIATION OF GAAP TO NON-GAAP FINANCIAL MEASURES
(U.S. dollars in thousands, except per share data)
(Unaudited)
Nine months ended
September 30
Three months ended
September 30
2024
2023
2024
2023
GAAP operating expenses:
$
12,275
$
11,886
$
4,274
$
4,367
Change of operating lease accounts
(8)
47
6
14
Share-based compensation to employees, directors
and consultants
(1,295)
(1,373)
(515)
(521)
Non-GAAP operating expenses:
10,972
10,560
3,765
3,860
GAAP operating loss
(13,277)
(2,444)
(4,542)
(4,602)
Change of operating lease accounts
8
(47)
(6)
(14)
Share-based compensation to employees, directors
and consultants
1,295
1,373
515
521
Non-GAAP operating loss
(11,974)
(1,118)
(4,033)
(4,095)
GAAP Net loss
(12,731)
(2,330)
(4,326)
(4,377)
Change of operating lease accounts
(61)
(282)
41
(101)
Share-based compensation to employees, directors
and consultants
1,295
1,373
515
521
Non-GAAP Net loss
$
(11,497)
$
(1,239)
$
(3,770)
$
(3,957)
GAAP basic and diluted loss per ordinary share
$
(1.11)
$
(0.20)
$
(0.38)
$
(0.38)
NON- GAAP basic and diluted loss per ordinary share
$
(1.0)
$
(0.11)
$
(0.33)
$
(0.35)
About CollPlant
CollPlant is a regenerative and aesthetic medicine company focused on 3D bioprinting of tissues and organs, and medical aesthetics. The Company’s products are based on its rhCollagen (recombinant human collagen) produced with CollPlant’s proprietary plant-based genetic engineering technology. These products address indications for the diverse fields of tissue repair, aesthetics, and organ manufacturing, and are ushering in a new era in regenerative and aesthetic medicine.
In 2021, CollPlant entered into a development and global commercialization agreement for dermal and soft tissue fillers with Allergan, an AbbVie company, the global leader in the dermal filler market.
For more information about CollPlant, visit http://www.collplant.com
Use of Non-US GAAP (“non-GAAP”)
Financial results for 2024 and 2023 are presented on both a GAAP and a non-GAAP basis. GAAP results were prepared in accordance with U.S. GAAP and include all revenue and expenses recognized during the period. The release contains certain non-GAAP financial measures for operating costs and expenses, operating income (or loss), net income (or loss) and basic and diluted net income (or loss) per share that exclude the effects of non-cash expense for share-based compensation to employees, directors and consultants, and change in operating lease accounts. CollPlant’s management believes that these non-GAAP financial measures provide meaningful supplemental information regarding the Company’s performance that enhances management’s and investors’ ability to evaluate the Company’s operating costs, net income (or loss) and income (or loss) per share, and to compare them to historical Company results.
The presentation of this non-GAAP financial information is not intended to be considered in isolation or as a substitute for the financial information prepared and presented in accordance with GAAP. Management uses both GAAP and non-GAAP measures when operating and evaluating the Company’s business internally and therefore decided to make these non-GAAP adjustments available to investors. The non-GAAP financial measures used by the Company in this press release may be different from the measures used by other companies.
For more information on the non-GAAP financial measures, please see the “Reconciliation of GAAP to Non-GAAP Financial Measures” in this release. This accompanying table has more details on the GAAP financial measures that are most directly comparable to non-GAAP financial measures and the related reconciliations between these financial measures.
The Company’s consolidated financial statements for the third quarter ended September 30, 2024, are presented in accordance with generally accepted accounting principles in the U.S.
Forward-Looking Statements
This press release may include forward-looking statements. Forward-looking statements may include, but are not limited to, statements relating to CollPlant’s objectives plans and strategies, as well as statements, other than historical facts, that address activities, events or developments that CollPlant intends, expects, projects, believes or anticipates will or may occur in the future. These statements are often characterized by terminology such as “believes,” “hopes,” “may,” “anticipates,” “should,” “intends,” “plans,” “will,” “expects,” “estimates,” “projects,” “positioned,” “strategy” and similar expressions and are based on assumptions and assessments made in light of management’s experience and perception of historical trends, current conditions, expected future developments and other factors believed to be appropriate.
Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements. Many factors could cause CollPlant’s actual activities or results to differ materially from the activities and results anticipated in forward-looking statements, including, but not limited to, the following: the Company’s history of significant losses, its need to raise additional capital and its inability to obtain additional capital on acceptable terms, or at all, including uncertainties surrounding the methods of fundraising and the Company’s preferences regarding such methods; the Company’s expectations regarding the costs and timing of commencing and/or concluding pre-clinical and clinical trials with respect to breast implants, tissues and organs which are based on its rhCollagen based BioInk and other products for medical aesthetics, and specifically the Company’s ability to initiate its next large-animal study for its breast implants in a timely manner, or at all; the Company’s or Company’s strategic partners’ ability to obtain favorable pre-clinical and clinical trial results; regulatory action with respect to rhCollagen-based bioink and medical aesthetics products or product candidates including, but not limited to, acceptance of an application for marketing authorization review and approval of such application, and, if approved, the scope of the approved indication and labeling; commercial success and market acceptance of the Company’s rhCollagen based products, in 3D Bioprinting and medical aesthetics; the Company’s ability to establish sales and marketing capabilities or enter into agreements with third parties and its reliance on third party distributors and resellers; the Company’s ability to establish and maintain strategic partnerships and other corporate collaborations, including its partnership with AbbVie and its ability to continue to receive milestone and royalties payments under the AbbVie agreement; the Company’s reliance on third parties to conduct some or all aspects of its product development and manufacturing; the scope of protection the Company is able to establish and maintain for intellectual property rights and the Company’s ability to operate its business without infringing the intellectual property rights of others; current or future unfavorable economic and market conditions and adverse developments with respect to financial institutions and associated liquidity risk; the impact of competition and new technologies; general market, political, and economic conditions in the countries in which the Company operates, including, with respect to the ongoing war in Israel, projected capital expenditures and liquidity, changes in the Company’s strategy and development plans and projects,, and litigation and regulatory proceedings. More detailed information about the risks and uncertainties affecting CollPlant are contained under the heading “Risk Factors” included in CollPlant’s most recent annual report on Form 20-F filed with the SEC, and in other filings that CollPlant has made and may make with the SEC in the future. The forward-looking statements contained in this press release are made as of the date of this press release and reflect CollPlant’s current views with respect to future events, and CollPlant does not undertake and specifically disclaims any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Contacts
CollPlant:
Eran Rotem
Deputy CEO & CFO
Tel: + 972-73-2325600
Email: Eran@collplant.com
Investors:
LifeSci Advisors
Dan Ferry
daniel@lifesciadvisors.com
Photo – https://mma.prnewswire.com/media/2568075/CollPlant.jpg
Logo – https://mma.prnewswire.com/media/2217353/CollPlant_Logo.jpg
View original content to download multimedia:https://www.prnewswire.com/news-releases/collplant-biotechnologies-reports-2024-third-quarter-financial-results-and-provides-corporate-update-302317380.html
SOURCE CollPlant
You may like
Technology
Truck Accident Attorney Network Relaunches Website to Expand Nationwide Visibility and Elite Truck Accident Lawyers
Published
43 minutes agoon
April 20, 2026By
LOS ANGELES, April 20, 2026 /PRNewswire/ — Truck Accident Attorney Network is proud to announce the official relaunch of its newly redesigned website https://www.truckaccidentattorneynetwork.org/, making it a major step forward in its mission to connect accident victims with highly qualified, experienced truck accident lawyers across the United States.
The updated platform features a modern design, an improved user experience, and enhanced functionality to better serve individuals seeking legal representation after serious truck accidents. With a renewed focus on nationwide growth, Truck Accident Attorney Network aims to expand its reach like never before to help people get top-performing attorneys who specialize in complex truck accident litigation.
Unlike other traditional legal directories, Truck Accident Attorney Network aims to implement a strict vetting process to ensure that only highly experienced attorneys are included. All the candidates that are considered must meet the organization’s rigorous membership standards, which can be reviewed here: https://www.truckaccidentattorneynetwork.org/membership-criteria/.
“Unlike many legal directories, attorneys can’t just sign up and advertise on our website. Every attorney must meet our membership criteria and demonstrate experience handling truck accident cases. The purpose of the Truck Accident Attorney Network is to ensure injured victims can connect with qualified truck accident lawyers, not just any personal injury attorney. Our goal is simple — when someone finds a lawyer through https://www.truckaccidentattorneynetwork.org/, they know they are being connected with an elite attorney who has real experience handling serious truck accident cases.”
To prioritize quality over quantity, Truck Accident Attorney Network ensures that only attorneys who possess the track record and expertise needed to tackle high-stakes truck accident claims are available to choose from. The relaunch reinforces the website’s commitment to transparency, trust, and results – driven legal connections.
For more information, visit https://www.truckaccidentattorneynetwork.org/.
View original content:https://www.prnewswire.com/news-releases/truck-accident-attorney-network-relaunches-website-to-expand-nationwide-visibility-and-elite-truck-accident-lawyers-302747839.html
SOURCE Everest Legal Marketing
Technology
Trutankless® Expands GEN3 Lineup with 208V Unit Built for Condos, Multi-Family, and Light Commercial Use
Published
43 minutes agoon
April 20, 2026By

Engineered for 208V environments, the new unit delivers a compact, energy-efficient, low-maintenance alternative to traditional tank water heaters
SCOTTSDALE, Ariz., April 20, 2026 /PRNewswire/ — Trutankless® today announced the release of its GEN3 Commercial 208V Unit, now in stock and available through wholesalers nationwide, bringing its award-winning “Smart” technology – trusted in homes across the country – into light commercial environments for the first time.
Built for environments like salons, restaurants, fitness studios, and multi-unit properties, the GEN3 208V unit gives business owners a simple, reliable solution: consistent hot water, no downtime, and less maintenance.
A Smarter Upgrade for Commercial Spaces
Many commercial and residential buildings already operate on 208-volt electrical systems, making the GEN3 unit an easy transition from bulky, high-maintenance tanks to a more modern, energy-efficient solution.
With a compact, wall-mounted design and advanced internal controls, the system is designed to perform under pressure, especially during peak business hours when hot water demand is highest.
Key Features & Smart Capabilities Include:
Precision Temperature Control
Advanced solid-state electronics continuously modulate power to maintain consistent water temperature within a fraction of a degree, even during peak usage times.
Low-Maintenance, Long-Life Design
A proprietary heat exchanger is engineered to resist scale and mineral buildup, helping extend the system’s lifespan and significantly reduce the maintenance typically required with traditional tank heaters.
Smart Monitoring & Proactive Maintenance
Integrated Wi-Fi connectivity transforms hot water management from a reactive task into a proactive strategy. Through a centralized digital dashboard, property owners and facility managers gain a comprehensive view of system performance across one or multiple units.
Proactive System Health – Predictive diagnostics help identify potential issues before they lead to downtime.Preventative Oversight – Real-time status updates and maintenance reminders help ensure systems operate at peak reliability.Comprehensive Dashboard – Monitor performance metrics, track energy usage, and manage multiple units from a single interface.Smart Alerts – Instant notifications enable faster troubleshooting and help minimize service interruptions.
Space-Saving Installation
The sleek, wall-mounted design frees up valuable floor space, giving businesses more room for operations, storage, or customer-facing areas.
Energy-Efficient Operation
By eliminating standby heat loss, the GEN3 unit helps reduce overall energy consumption, lowering utility costs while also supporting a smaller environmental footprint.
Meeting Growing Demand for Electric Solutions
The launch comes as more businesses look for efficient, electric-first infrastructure that is easier to manage and more cost-effective over time.
“The feedback from our partners and early adopters has been nothing short of spectacular,” said Guy Newman, CEO of Trutankless®. “We’ve seen a massive surge in demand for a high-performance 208V solution that doesn’t compromise on reliability. The GEN3 Commercial unit is the culmination of years of engineering – it’s smarter, tougher, and more efficient than anything else on the market.”
Available Now Through Wholesale Partners
The GEN3 Commercial 208V unit is now in stock and available through authorized Trutankless® wholesale partners nationwide.
For more information or to find a local distributor, visit www.trutankless.com.
About Trutankless®
Trutankless® is a leading innovator in electric tankless water heating technology, focused on delivering high-performance, energy-efficient solutions for residential and commercial applications. Based in Arizona, the company continues to push the industry forward with smart, space-saving systems designed for modern living and working environments.
View original content to download multimedia:https://www.prnewswire.com/news-releases/trutankless-expands-gen3-lineup-with-208v-unit-built-for-condos-multi-family-and-light-commercial-use-302747862.html
SOURCE Trutankless
Technology
LITO Announces New Collaboration Bringing Exclusive Contemporary Artist Editions to Sotheby’s Online Marketplace
Published
44 minutes agoon
April 20, 2026By

The initiative sees artists create new works designed from the outset for high-quality, limited editions.
NEW YORK, April 20, 2026 /PRNewswire/ — LITO, a pioneering printmaking company redefining how art is created and collected, is pleased to announce a new collaboration that brings exclusive, museum-quality contemporary art editions to Sotheby’s online marketplace. The collaboration introduces a curated program of LITO Editions by leading international artists, offering collectors a new way to acquire highly refined, limited-edition works conceived in direct collaboration with the artists themselves.
LITO Editions are authentic works of art conceived in direct collaboration with artists, who imagine original work specifically for the format. The pieces are then developed into limited editions at LITO’s Technology Lab, an in-house research and production facility where engineers, technicians, and print specialists have developed the company’s patented Hi-Rnd© process. The lab brings together precision engineering and artistic collaboration to produce works that capture texture, color, relief, and brushstroke detail at multiple scales. Produced in limited runs, the editions are each hand-signed and numbered by the artist.
The initiative begins with an exclusive series of LITO Editions by acclaimed American portrait artist Kehinde Wiley. The series is based on his 2008 painting Triple Portrait of Charles II. Drawing on the historical tradition of multi-view portraiture, these works explore identity as layered and constructed rather than singular. Each piece is produced as a high-resolution print on Aludibond, set within an aluminum frame. The compositions feature engraved floral elements against mirrored backgrounds, available in Gold, Dark Mirror, and Mirror finishes, with select works also offered in a blue variant. Available for immediate purchase on Sotheby’s online marketplace, the editions are priced between $8,000 and $15,000, expanding access to Wiley’s work while maintaining the highest standards of craftsmanship.
Wiley’s editions are currently on view at Sotheby’s galleries in the Breuer Building in New York through April 24, offering collectors and the public an opportunity to experience the artist’s work and LITO’s high-resolution editions firsthand. The presentation highlights the depth, texture, and dimensionality that define LITO’s approach and underscores the collaboration’s emphasis on in-person engagement.
“The marriage of art and science is one that’s been known and storied. There was, prior to the camera, an assumption that art was the ultimate authority of truth in history. Now with new technology, art is freed to take on new responsibilities, and to be able to tell other types of stories,” said Wiley. “Working with Sotheby’s, LITO, and LITO’s technology has also allowed me to go back into my archive and rethink paintings that had been done years ago and these editions are part of a grand tradition of artists playing with the leading edge of technology.”
“As the art market continues to evolve, the collaboration signals a broader shift toward new formats and technologies that expand how art is created, distributed, and collected, without compromising on artistic intent or quality,” said John Dodelande, CEO of LITO. “By creating a new asset class within the art market, our technology will allow a new generation of collectors to experience and immerse themselves in the art world in a way that limited supply may have otherwise precluded them from.”
“Beginning a collection with editions offers an exciting and accessible entry point into contemporary art,” said Cynthia Houlton, Sotheby’s Senior Vice President and Global Head of Demand Generation & Marketplace. “Through our collaboration with LITO, collectors can acquire high‑quality works directly from the artist, reinforcing both authenticity and a meaningful connection to the creative process. Exclusive to Sotheby’s, these editions carry a compelling sense of rarity while remaining approachable. By presenting them on our Marketplace, we aim to welcome new audiences and invite discovery of exceptional art in a way that feels both inspiring and inclusive.”
New releases and exhibitions will follow throughout 2026, with monthly drops planned alongside presentations in New York, Los Angeles and London, including a Sotheby’s showcase in London from April 29 to May 17 and in Beverly Hills from May 18 to May 31.
Founded in 2022 and based in Bregenz, Austria, LITO already operates showrooms in Paris, London, and Bregenz, reflecting its growing international footprint. The company has collaborated with a wide range of internationally recognized contemporary artists, including Amoako Boafo, Daniel Arsham, Camille Henrot, Jia Aili, and Peter Halley, among others, advancing a model that creates new pathways for artists while extending the reach and accessibility of their work.
ABOUT LITO:
Founded in 2022, LITO is a forward-thinking printmaking company based in Bregenz, Austria. Through its patented Hi-Rnd© technology, LITO produces high-rendered limited editions that capture the texture, color, and dimensionality of original artworks with exceptional precision. Working in close collaboration with leading contemporary artists, LITO creates editions that are hand-signed, numbered, and conceived specifically for this innovative format.
ABOUT SOTHEBY’S
Established in 1744, Sotheby’s promotes access and ownership of exceptional art and luxury objects through auctions, private sales and retail. Our deep expertise across 70 selling categories is supported by a leading technology platform and a global network of specialists spanning 40 countries. Selling categories include Contemporary Art, Modern and Impressionist Art, Old Masters, Chinese Works of Art, Jewelry, Watches, Wine and Spirits and Design, as well as collectible cars and real estate through RM Sotheby’s and Concierge. Sotheby’s Financial Services is a leading art lender and provides capital solutions for collectors around the world, having originated more than $12 billion in loans since its inception. Sotheby’s new global headquarters is now open at the iconic Breuer building at 945 Madison Avenue in New York City.
PRESS CONTACTS:
For LITO: Ashley Hansen, Forward Global, ashley.hansen@forwardglobal.com
For LITO: Kyle Boulia, Forward Global, kyle.boulia@forwardglobal.com
For LITO: Sloan Savage, Forward Global, sloan.savage@forwardglobal.com
View original content to download multimedia:https://www.prnewswire.com/news-releases/lito-announces-new-collaboration-bringing-exclusive-contemporary-artist-editions-to-sothebys-online-marketplace-302747855.html
SOURCE LITO Editions

Truck Accident Attorney Network Relaunches Website to Expand Nationwide Visibility and Elite Truck Accident Lawyers

Trutankless® Expands GEN3 Lineup with 208V Unit Built for Condos, Multi-Family, and Light Commercial Use

LITO Announces New Collaboration Bringing Exclusive Contemporary Artist Editions to Sotheby’s Online Marketplace

Whiteboard Series with NEAR | Ep: 45 Joel Thorstensson from ceramic.network
Send Rakhi to UK swiftly with UK Gifts Portal

New Gooseneck Omni Antennas Offer Enhanced Signals in a Durable Package

Why You Should Build on #NEAR – Co-founder Illia Polosukhin at CV Labs

Whiteboard Series with NEAR | Ep: 45 Joel Thorstensson from ceramic.network

NEAR End of Year Town Hall 2021: The Open Web World, MetaBUILD 2 Hackathon and 2021 recap
Trending
-

 Technology3 days ago
Technology3 days agoInterfaith America Works to Promote Free, Fair and Peaceful Elections
-

 Coin Market3 days ago
Coin Market3 days agoFrench finance minister backs euro-pegged stablecoins to compete with US
-

 Technology1 day ago
Technology1 day agoHarmonic Enables DIRECTV to Reimagine Nationwide DTH Service
-

 Near Videos3 days ago
Near Videos3 days agoWe Have Only Scratched The Surface Of The Agentic Future
-

 Coin Market3 days ago
Coin Market3 days agoSingapore Gulf Bank adds stablecoin mint and redeem for 24/7 settlement
-

 Near Videos3 days ago
Near Videos3 days agoAnthropic Cuts Off OpenClaw Subscribers | GPT-Image-2 Leaked | Drift $285M Hack Explained
-

 Near Videos3 days ago
Near Videos3 days agoNEAR Intern Demos the Future of Private Trading
-

 Technology3 days ago
Technology3 days agoDynamite Integrates Biometric Cryptography and AI into its Wallet Product

